Probiotics are defined as “Live microorganisms, which when administered in adequate amounts, confer a health benefit on the host”[1] (a.k.a., friendly bacteria). In any case, there is no question that probiotics are a trending dietary supplement category. Of course, various factors are driving probiotic supplement manufacturing trends and innovation opportunities, which I’ll cover in this article.
Global Overview and Size of the Probiotics Supplement Market
According to an International Food Information Council survey[2] of 1,000 adults, nearly one in four people say that digestive health is the most important aspect of their overall health. Over half consider themselves familiar with probiotics, although fewer are familiar with postbiotics and synbiotics…more on these later), and one in three actively try to consume them. Interestingly, 44% of people using probiotics seek them in supplements, while 54% seek them in foods like yogurt.
The global probiotics market size was estimated at $77.12 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 14.0% from 2023 to 2030.[3] This expansion is a key indicator for businesses, investors, and industry analysts, guiding market analysis and strategic planning. Keep reading to discover the top factors fueling the growth of the probiotics supplement market.

What Probiotic Strains Can Be Used in Dietary Supplements?
While many probiotic strains could be utilized in a dietary supplement, the best choices are those probiotic strains whose benefits are supported by human clinical research AND which have good stability—meaning that they will both maintain their potency in the bottle for a reasonable period and, once ingested, make it through to the gut without being largely destroyed by stomach acids.
Examples of good choices include spore-forming probiotics, such as those in the Bacillus genus. Spore-forming probiotic bacteria possess a protective encasement that allows almost indefinite storage until it is ready to be consumed and can survive transit through the stomach intact.
There are other select probiotic bacteria in the Lactobacillus and Bifidobacterium genera, which are relatively hardy and can survive to deliver their various health benefits. A good contract probiotic supplement manufacturer will formulate probiotic products with intentional potency overages based on the survivability of each strain used. This is important, as the FDA requires dietary supplements to meet 100% of label claims until the expiration date.
What Factors Are Driving the Growth of the Probiotics Supplement Market?
The growth of the probiotic supplement market during the last two decades may be attributed to the continuously expanding scientific evidence pointing to their beneficial effects on human health.[4]
The top reasons provided for using probiotics are gut health, general health and wellness, and immune health. People under age 45 were most likely to say they consume probiotics to support their mental health.[5]An emerging growth area in the probiotic supplement market is the increased availability and consumer awareness of postbiotics and synbiotics.
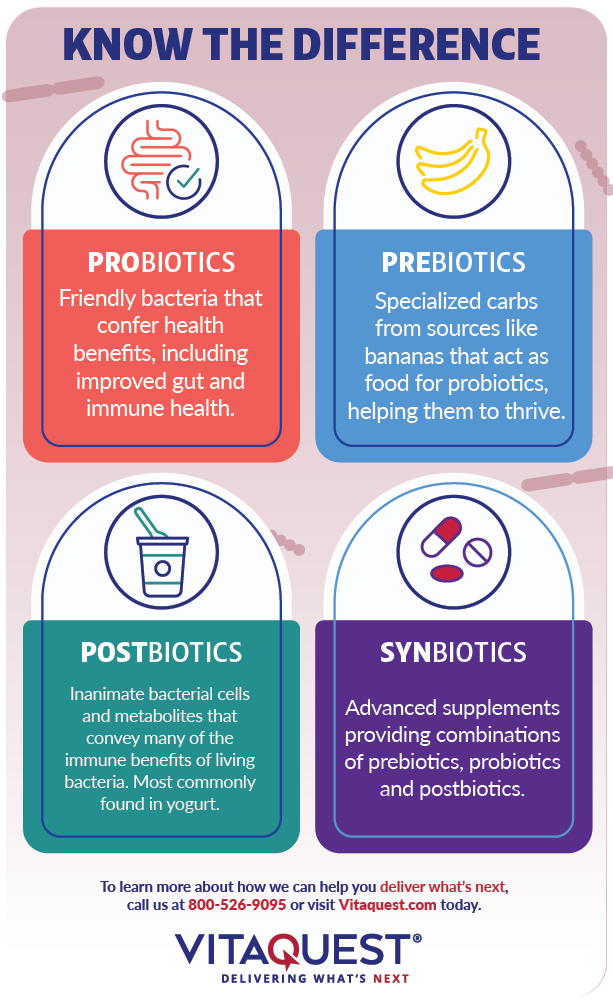
Postbiotics are not alive. Rather, they are the end-products of probiotic bacteria, including inanimate bacterial cells (which can still convey many of the immune benefits of living bacteria) and bacterial metabolites (such as short-chain fatty acids). The most common example of postbiotics is found in yogurt, where the active bacterial cultures produce the beneficial lactic acid that gives yogurt its refreshing taste while helping to promote healthy colonic and vaginal pH.
Synbiotics are supplements specially formulated to provide probiotics in combination with prebiotics. Prebiotics are specialized carbohydrates found in foods such as chicory root, Jerusalem artichoke, and bananas, which are ideal for gut bacteria. In this way, your colonic bacteria convert prebiotics into postbiotics.
Considerations for Formulating and Manufacturing Probiotic Supplements
Another important consideration is the actual process of manufacturing probiotic supplements. First and foremost, when probiotics arrive at the manufacturing facility, they must be placed in appropriate storage conditions to guard against potency loss.
For most probiotic ingredients, this means large walk-in coolers maintained at tightly controlled temperatures under 40°F (4°C). Some bacterial strains require freezer temperatures, yet other ingredients, such as prebiotics and postbiotics, might require controlled room temperature, so the ideal environment for each incoming ingredient must be carefully chosen.
Secondly, extra care must be taken in cleaning the manufacturing rooms, equipment, and utensils before each production run to ensure that no ingredient cross-contamination takes place. For the same reason, air quality should be routinely monitored. This is especially important for probiotics since they don’t always play well with all other nutraceuticals.
For those of you reading this article who might be thinking about creating a product, it is important to select a probiotics supplement manufacturer that is not only capable of producing these products, but also has experience in how to handle probiotic strains, understands which ones have stability and will survive stomach acids and bile to ultimately provide their benefits, and which delivery forms work best for probiotics.
Innovation Opportunities in Manufacturing Probiotics Supplement
While gut/digestive health and immune health are categories of focus for probiotics, there’s a wide range of specific probiotic strains that present opportunities for innovation.
For instance, clinical trials have demonstrated that a strain of Lactobacillus gasseri can significantly reduce visceral fat[6]. Moreover, strains such as Lactobacillus fermentum, Lactobacillus rhamnosus, Lactobacillus plantarum, and Bifidobacterium longum have been shown to promote sleep quality. Additionally, a specific strain of Bifidobacterium longum was shown to help improve skin gloss and firmness and reduce redness, among other benefits. These are just a few examples of the diverse benefits various probiotic strains offer.
Other opportunities for probiotic innovation involve using delivery systems that protect probiotic strains from destruction by stomach acid. This includes microencapsulation technology for individual probiotic strains, and enteric coating (and similar coatings) for finished capsules and tablets.
Given the exponential growth of this category, which is being fueled even more by the introduction and growing acceptance of postbiotic and synbiotic products, it is important to consider innovation opportunities to give you a point of differentiation when introducing products in this dynamic category.
Why Choose Vitaquest as Your Probiotic Supplement Manufacturing Company?
Vitaquest, a reputable contract manufacturer with over 45 years of experience, specializes in manufacturing probiotic supplements. Leveraging this extensive experience, Vitaquest offers guidance and technical expertise to ensure your probiotic product meets or surpasses the standards for cutting-edge supplements, with a keen focus on probiotic supplement trends. In addition to world-class manufacturing, Vitaquest provides expert supplement packaging services, best-in-class particle and powder engineering services, and convenient fulfillment and warehousing solutions.
In addition, Vitaquest continues to invest in specialized capabilities to support the evolving demands of probiotic supplement manufacturing. In February 2026, Vitaquest opened a dedicated Probiotics Manufacturing Suite designed specifically for handling live cultures. This specialized space allows for greater control during production, helping protect probiotic stability and potency while supporting a wider range of formulations. Combined with in-house ingredient preparation capabilities, Vitaquest offers a streamlined approach for developing high-quality probiotic supplements with consistency and reliability.
Discover how we can help you enhance or create a high-quality probiotic supplement. Call 800-526-9095 to speak with one of our knowledgeable sales executives or request a competitive nutraceutical manufacturing price quote for this service.
Frequently Asked Questions about Probiotic Supplement Manufacturing Trends
What is the probiotic supplement market worth?
The global probiotics market size was estimated at $77.12 billion in 2022.
What is the future of probiotics?
The future of probiotics is bright. The market is expected to grow at a compound annual growth rate (CAGR) of 14.0% from 2023 to 2030.
What are the challenges in the production of probiotics?
The contract probiotic supplement manufacturer needs to store the probiotics correctly, formulate with sufficient potency overages to guarantee shelf life, and make sure to combine probiotics only with those ingredients that won’t negatively impact their potency. For example, anti-bacterial ingredients such as citrus oils may also kill good bacteria such as probiotics.
What are the reasons for the increasing popularity of probiotic products in short?
The growth of the probiotic supplement market during the last two decades may be a result of the continuously expanding scientific evidence pointing to their beneficial effects on human health.
What factors can influence the effectiveness of probiotics?
A big issue is stability. Will the probiotic strains survive stomach acids and bile? How will the strain hold up over time? A good probiotic supplement manufacturer can help you choose hardy strains, and/or strains with a delivery system designed to protect them from destruction.
References
[1] FAO/WHO. Health and Nutritional Properties of Probiotics in Food including Powder Milk with Live Lactic Acid Bacteria. Report of a Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria; 2001.
[2] Consumer Insights on Gut Health & Probiotics. International Food Information Council. April 2022. Retrieved February 1, 2024 from chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://foodinsight.org/wp-content/uploads/2022/04/IFIC-Gut-Health-and-Probiotics-Survey.pdf.
[3] Probiotics Market Size, Share & Trends Analysis Report By Product (Food & Beverages, Dietary Supplements), By Ingredient (Bacteria, Yeast), By Distribution Channel, By End-use, By Region, And Segment Forecasts, 2023 – 2030. Grand View Research. Retrieved February 1, 2024 from https://www.grandviewresearch.com/industry-analysis/probiotics-market?utm_source=google&utm_medium=paid_ad&utm_campaign=cmfe_latest&utm_term=probiotics_market&gclid=Cj0KCQiAn-2tBhDVARIsAGmStVlJ75EcV-khujGUBx-eCLb4CiTc3Gzo0PK4d0ePV42srhwtKspfKi0aAhTbEALw_wcB
[4] Kechagia M, Basoulis D, Konstantopoulou S, Dimitriadi D, Gyftopoulou K, Skarmoutsou N, Fakiri EM. Health benefits of probiotics: a review. ISRN Nutr. 2013 Jan 2;2013:481651.
[5] Consumer Insights on Gut Health & Probiotics. International Food Information Council. April 2022. Retrieved February 1, 2024 from chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://foodinsight.org/wp-content/uploads/2022/04/IFIC-Gut-Health-and-Probiotics-Survey.pdf.
[6] Kim J, Yun JM, Kim MK, Kwon O, Cho B. Lactobacillus gasseri BNR17 Supplementation Reduces the Visceral Fat Accumulation and Waist Circumference in Obese Adults: A Randomized, Double-Blind, Placebo-Controlled Trial. J Med Food. 2018 May;21(5):454-461.